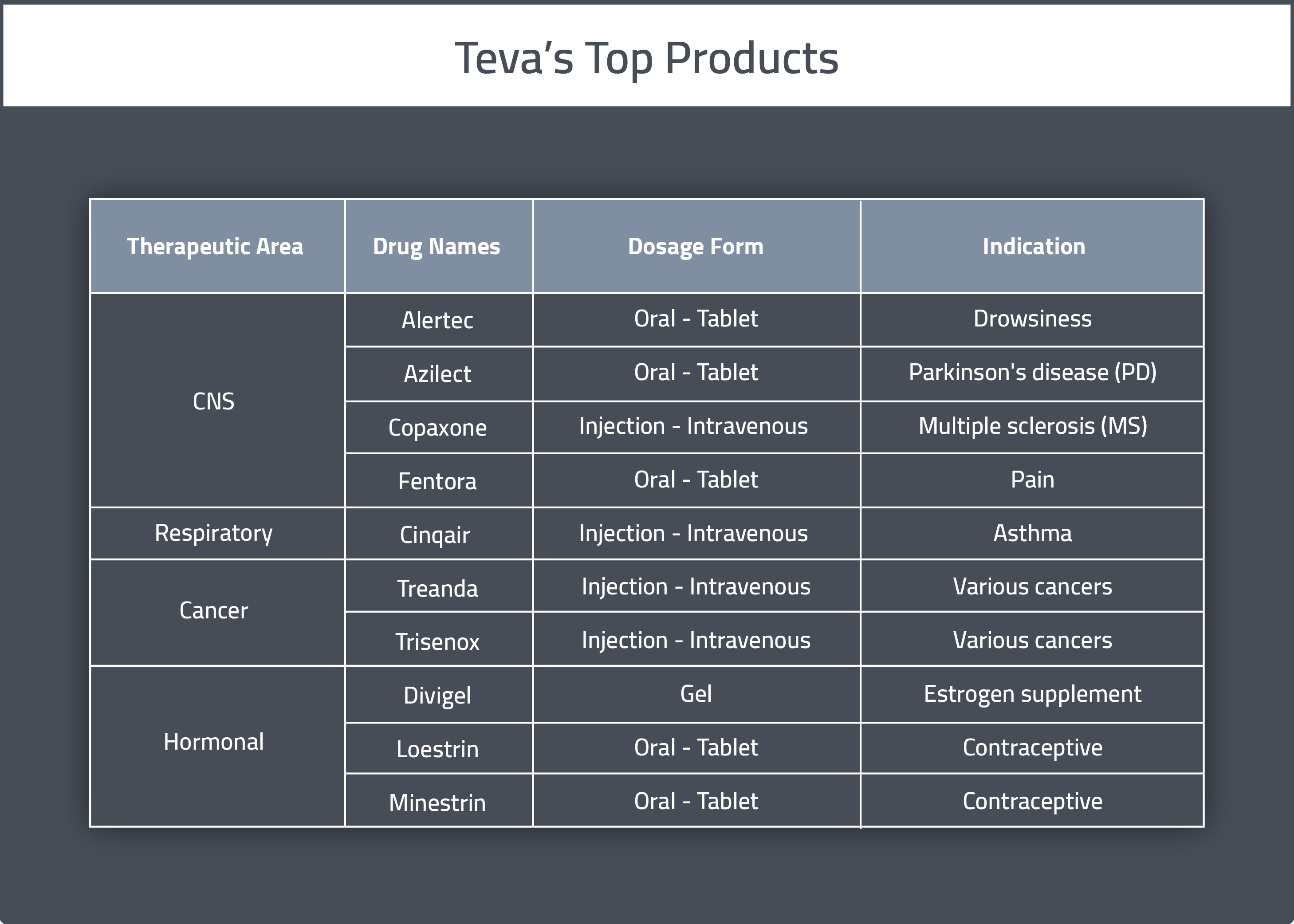
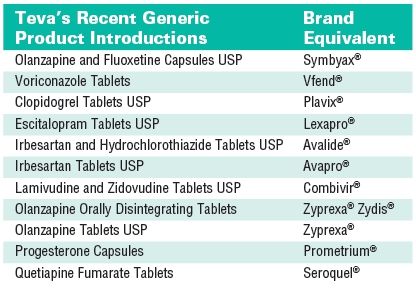
Huntington's disease drug: Teva Pharma settles patent dispute with Lupin, continues to litigate against Aurobindo

J&J, Teva offer a whopping $48B combined to wrap opioid suits, but not all plaintiffs are on board | Fierce Pharma

Teva Branded Pharmaceutical Products R&D;, . An electronic inhaler add-on module which store and transmit information related to inhaler use (eModule) PDR0000401 FCC ID 2AJVSPDR0000401

Out in Jan: 2nd Benchmark of pharma companies' actions on antibiotic resistance and access - Access to Medicine Foundation
UNITED STATES DISTRICT COURT SOUTHERN DISTRICT OF OHIO TAMMY HOPKINS, Plaintiff, Case No.: 20-675 v. JOHNSON & JOHNSON, JAN

The Procter & Gamble Company and Teva Pharmaceutical Industries Announce Formation of New Consumer Health Care Partnership | Procter & Gamble News