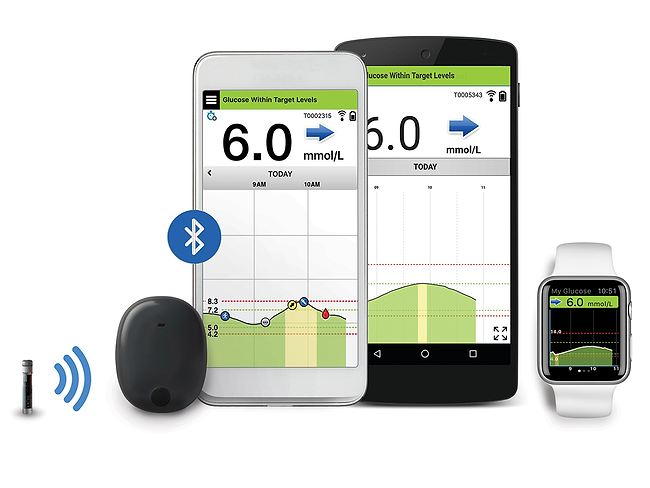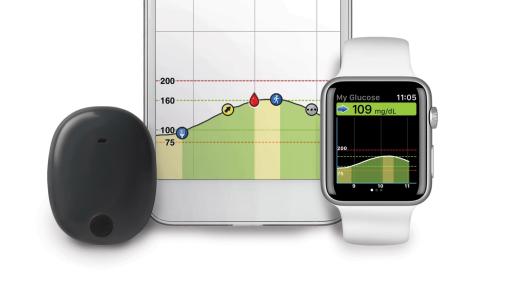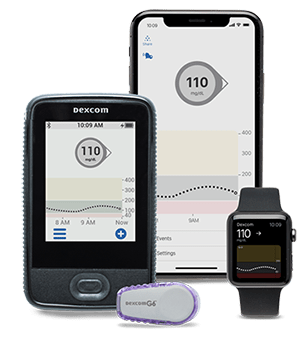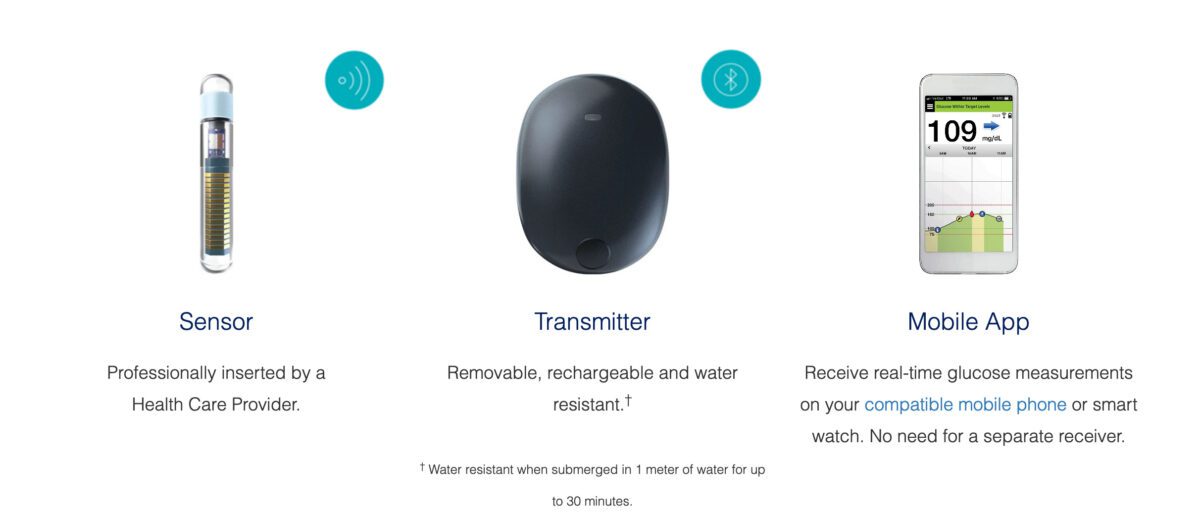
Eversense E3 CGM System for Up to 6 Months Receives FDA Approval Reports Senseonics – Medical Device News Magazine

FDA approves Eversense E3 6-month continuous glucose monitor that requires fewer fingerstick blood glucose measurements - NotebookCheck.net News