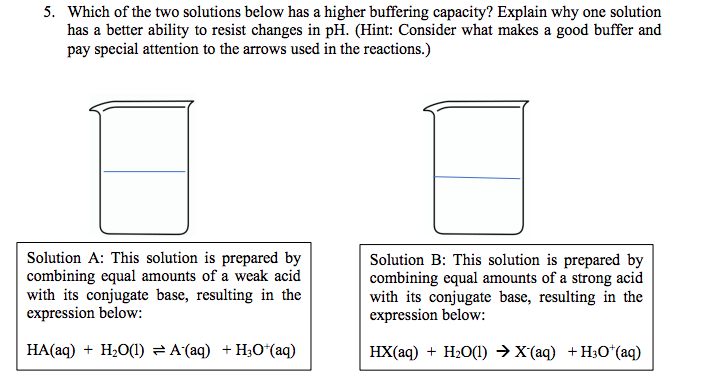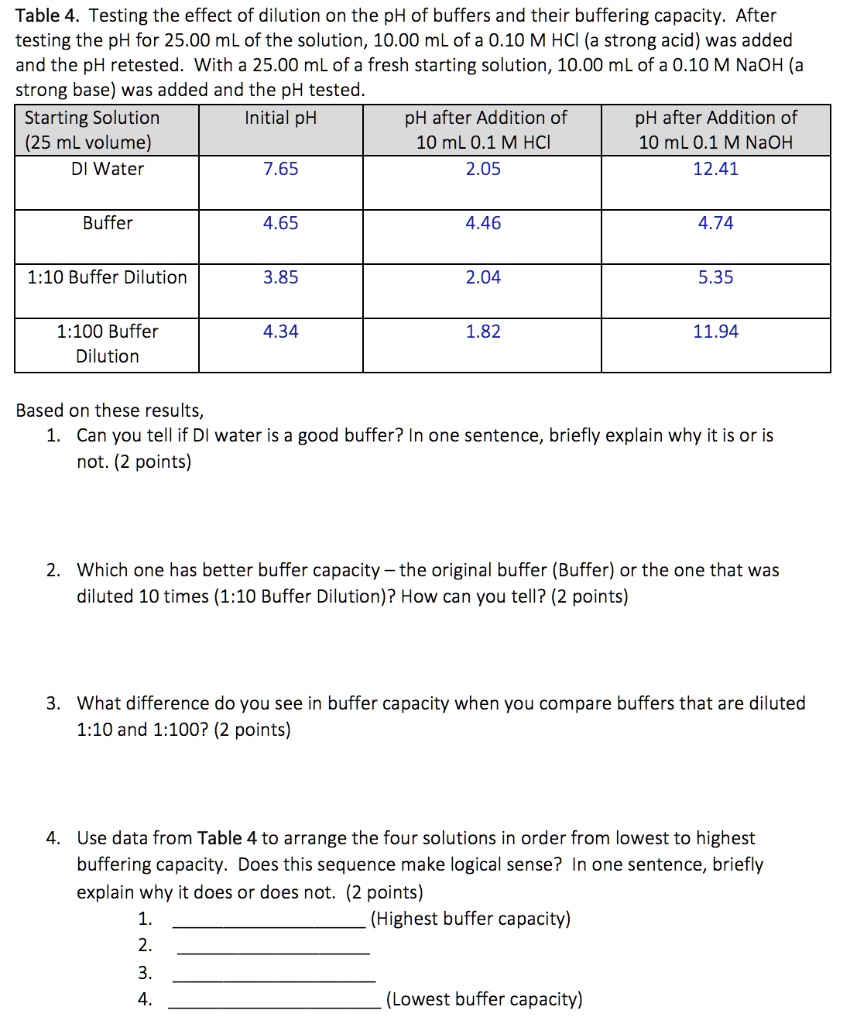
SOLVED:Table 4 Testing the effect of dilution on the pH of buffers and their buffering capacity: After testing the pH for 25.00 mL of the solution, 10.00 mLofa 0.10 M HCI (a

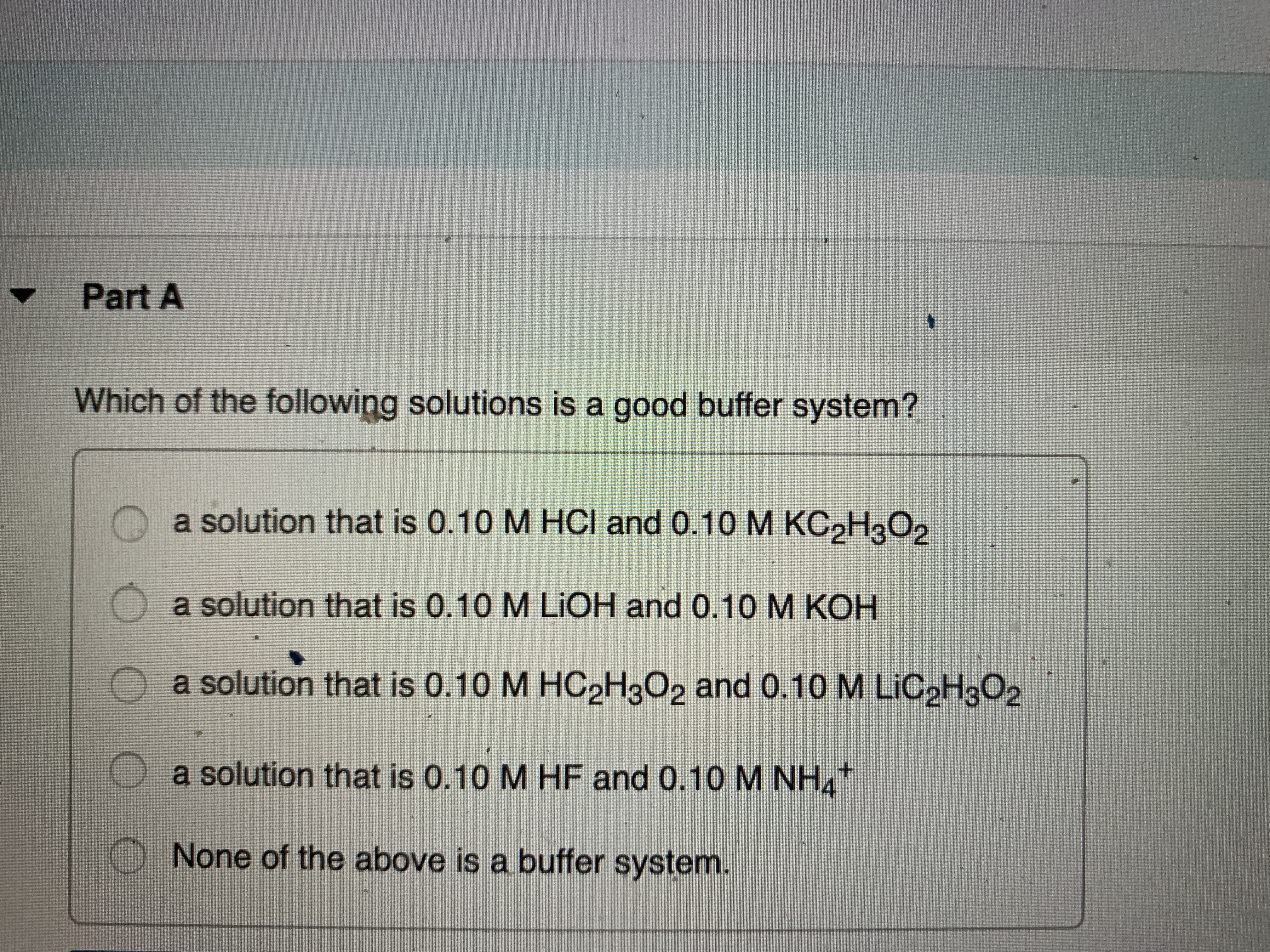
OneClass: 20) Identify a good buffer. A) small amounts of both a weak acid and its conjugate base B) ...







![Buffer vs Hootsuite Compared [2022]: Which is Better? - The Digital Merchant Buffer vs Hootsuite Compared [2022]: Which is Better? - The Digital Merchant](https://thedigitalmerchant.com/wp-content/uploads/2018/01/Buffer-vs-Hootsuite-Compared.png)


![10 Best Car Buffers 2020 [Buying Guide] – Geekwrapped 10 Best Car Buffers 2020 [Buying Guide] – Geekwrapped](https://uploads-ssl.webflow.com/5ad79663a6722a97c3d6a3f1/5ad79663a6722a3308d6e916_3_Best_Car_Buffers_Top_Image.png)